The pharmaceutical industry has long operated under a veil of biological neutrality. We often assume that a milligram of a miracle molecule behaves the same way in every vessel it enters. But as GLP-1 drugs like semaglutide and liraglutide move from niche diabetic treatments to cultural icons, a sharper reality is emerging. Modern research confirms that GLP-1 drugs affect men and women differently in ways that are both statistically significant and biologically profound.
Recent data suggests the gap is not merely anecdotal. It is written into the very architecture of our brains and our distinct hormonal cycles.
A groundbreaking study from the Icahn School of Medicine at Mount Sinai has finally provided the cartography we were missing. Published in Brain Medicine, the researchers constructed the first sex-specific atlas of GLP-1 expression. They found that the geography of these receptors is not uniform. In females, the brain regions responsible for appetite suppression are more densely populated with these docking stations. This explains why women frequently report a more profound silence in their hunger signals.
It is a matter of neurological density rather than a lack of willpower.
The researchers utilized a high-sensitivity technique called RNAscope to detect single mRNA transcripts. This allowed them to see what older methods missed. They mapped 25 distinct brain regions where GLP-1 sets up its biological home. The findings reveal that the hindbrain is the primary site of this divergence. In females, the density of GLP-1 neurons in the raphe obscurus nucleus and the solitary tract was significantly higher. These are the command centers for feeling full.
For men, the story shifts toward the senses. The study discovered that males have a much higher density of GLP-1 in the olfactory bulb. This is the region that processes smell. This suggests that for men, the drug may interact more heavily with the cephalic phase of metabolism. This is the stage where the mere scent of food triggers an insulin response. It is a fascinating pivot in how we understand the male metabolic experience.
This difference in brain geography manifests clearly in clinical outcomes.
A sweeping review recently published in JAMA Internal Medicine analyzed 64 clinical trials involving tens of thousands of patients. The findings were stark. On average, women lost approximately 11 percent of their starting body weight. Men in the same trials lost about 7 percent. While both figures represent a clinical success, the four percent margin is enough to change the conversation in a physician's office.
The market for these drugs is currently valued in the hundreds of billions.
We must consider the role of estrogen in this equation. The Mount Sinai team noted that estrogen appears to act as a biological amplifier for GLP-1 signals. In the female brain, estrogen receptors and GLP-1 neurons often sit in close proximity. They appear to be in a constant, coordinated conversation. This synergy may be why women experience a more potent suppression of appetite. It also suggests that as estrogen levels fluctuate, the efficacy of the drug might follow suit.
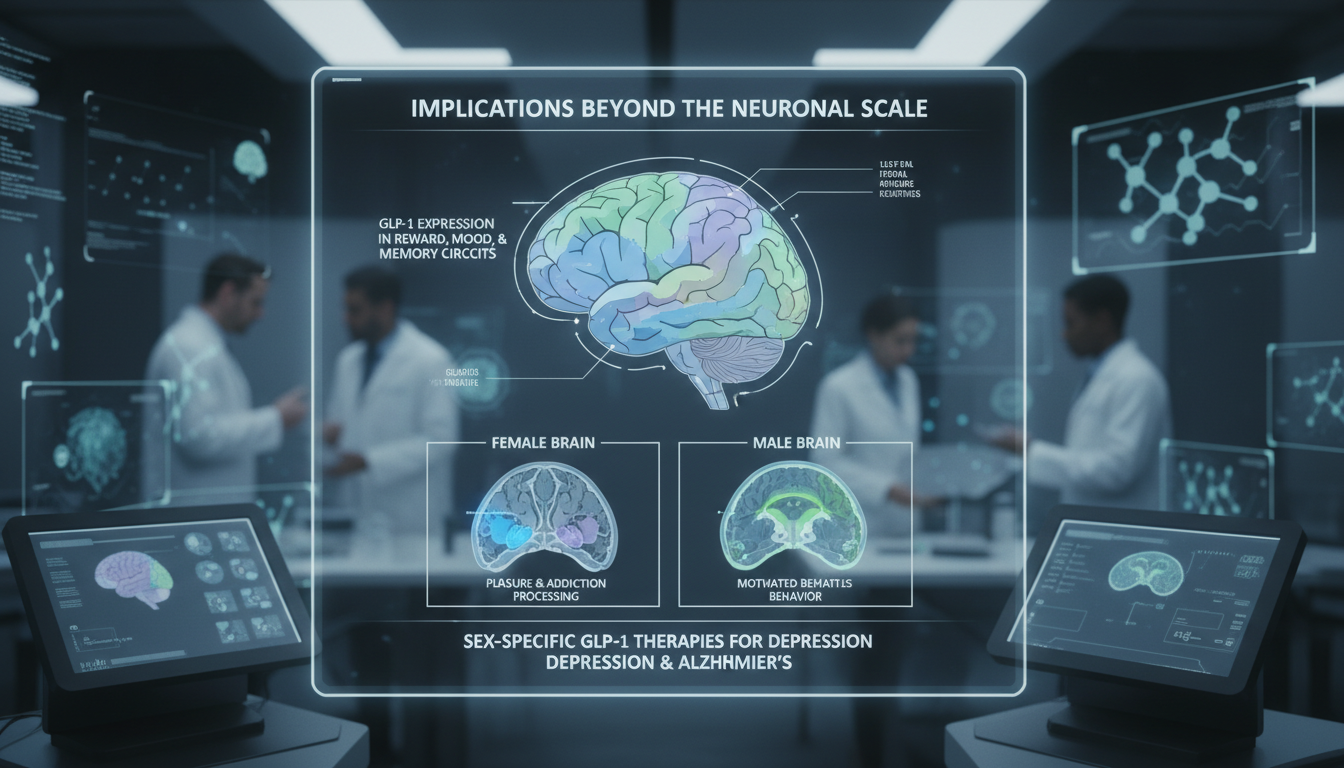
The implications of this atlas extend far beyond the scale. The researchers found GLP-1 expression in brain regions linked to reward processing, mood, and memory. In females, the ventral tegmental area showed distinct GLP-1 activity. This region is central to how we process pleasure and addiction. In males, the lateral hypothalamus, which governs motivated behavior, was the area of interest. This suggests that future GLP-1 therapies could be tailored by sex to treat depression or even Alzheimer’s disease.
This is the beginning of a move toward truly personalized metabolic medicine.
There is also the matter of sheer physical craft. Women generally have a lower median body weight and a different body composition than men. Since these drugs are often administered at fixed doses, a woman might simply be receiving a higher effective concentration of the molecule relative to her size. Furthermore, some experts speculate that women may be more adept at managing the gastrointestinal side effects that often lead to drug discontinuation.
The verdict is that biological sex is a primary driver of treatment success.
We are entering an era where the "one size fits all" approach to pharmacology is being dismantled. The Mount Sinai atlas and the JAMA meta-analysis both point toward a future where a prescription is adjusted not just for weight, but for the specific neural landscape of the patient. Understanding that a woman’s brain is literally wired to respond more intensely to these molecules is the first step in refining how we treat the modern metabolic crisis.
Frequently Asked Questions
Why do women lose more weight on GLP-1 drugs than men?
Research suggests women lose more weight because they have a higher density of GLP-1 receptors in the brain's appetite-control centers. Additionally, estrogen may enhance the drug's effects, and women often have a lower body weight which results in a higher relative dose.
Is Ozempic less effective for men?
It is not ineffective, but it is statistically less potent. Clinical reviews show men lose an average of 7 percent of their body weight compared to 11 percent for women. Men still see significant health benefits and metabolic improvements.
Can GLP-1 drugs help with brain-related issues like Alzheimer’s?
The latest brain mapping shows GLP-1 receptors in areas of the brain vulnerable to Alzheimer’s and those that control mood. Scientists believe these drugs could eventually be used to treat neuroinflammation, memory loss, and even addiction.
Does the smell of food affect men differently on these drugs?
Yes, the study found that men have more GLP-1 activity in the olfactory bulb, which handles smell. This suggests that for men, the drug may play a larger role in controlling the insulin spikes triggered by the scent of food.
Are the side effects of GLP-1 drugs different for men and women?
While both sexes experience similar side effects like nausea, some researchers believe women may be more resilient in managing these issues. This potentially leads to higher adherence rates and better long-term weight loss results for female patients.
How does estrogen interact with weight loss medications?
Estrogen appears to have a synergistic relationship with GLP-1. In the brain, estrogen can amplify the signal that tells the body it is full, making the medication feel more powerful for women than for men.
Was this study performed on humans?
The initial brain atlas was created using a murine (mouse) model because it allows for high-resolution mapping of brain tissues. However, the clinical weight loss data confirming the sex difference was gathered from 64 human clinical trials.



